Evidence of Endogenous DNA Synthesis In the Cytoplasm
Over the past years, there have been dispersed records of mammalian DNA observation in the cytoplasm, predominantly in terms of aging and disease. When you look at the DNA of a foreign organism, it is normal to find it in the cytoplasm, unlike the organism’s own genetic material, in the form of cDNA (complementary DNA) produced from an RNA strand, has mystified researchers. In all cases, the cDNA has emerged from endogenous retrotransposons, popular for their copy-and-paste method that leads to the addition of their own new copies into the genome. This mechanism usually occurs inside the nucleus, therefore, the cytoplasmic CDNA didn’t have an elucidation.

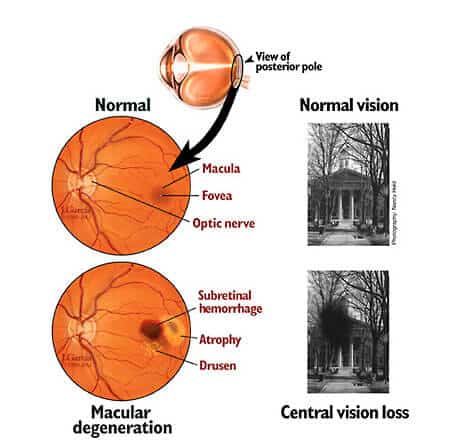
Jayakrishna Ambati, an ophthalmologist, and his teammates from the University of Virginia decided to explore the perplexing source after identifying the cDNA of Alu, the majorly found retrotransposons in the human genetic makeup, within the cell’s cytoplasm modeling a degenerative eye condition.
Their findings on the first evidence of DNA synthesis in the cytoplasm, which was published in PNAS on February 1 revealed that human cells can in fact create cDNA of Alu inside the cytoplasm.
Haig Kazazian, a geneticist from John Hopkins University who wasn’t a part of the study stated that this is significantly a pioneering finding.
Kazazian has researched retrotransposons for decades, concentrating particularly on L1 (Line-1). L1 codes for a protein with the capacity both to nick nucleic acids for the DNA to be incorporated and to synthesize RNA from DNA with reverse transcription. In the late 1990s, Kazazian and his team demonstrated that the copy-and-paste method of this actively mobile compound is coupled: the reverse transcription occurs at the genomic DNA cleavage site. L1 does this move to multiply itself in the genome, and Alu employs L1 to do the same.
Ambati’s research reveals that L1 is performing the reverse transcription of Alu within the cytoplasm, free of genomic insertion. Kazazian claimed that the discovery is provocative, and the proof backing it looks solid.
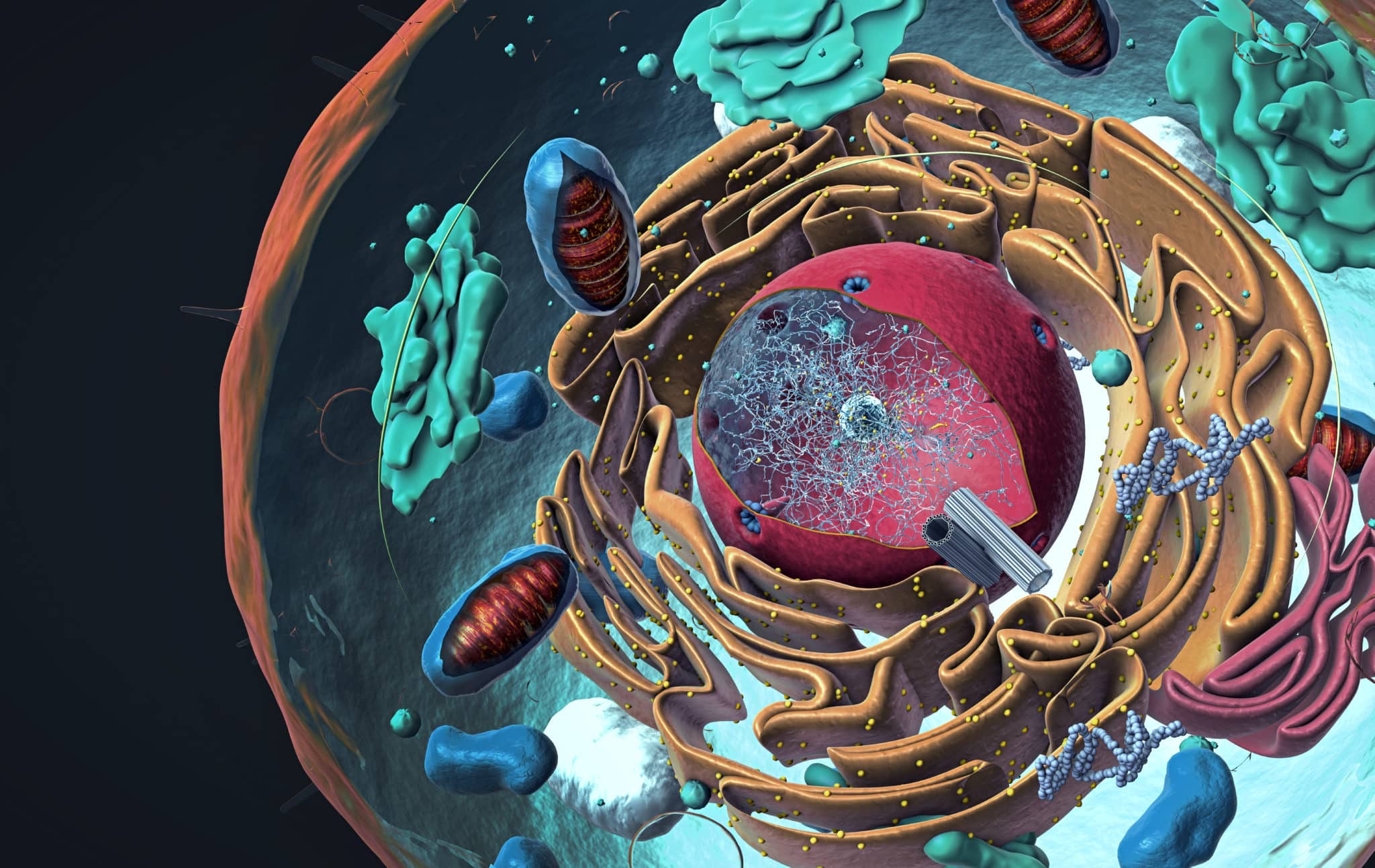
Starting DNA synthesis in the absence of genomic DNA presents a fascinating question: How this can occur in the absence of all the common equipment for the process? The enzyme, reverse transcriptases require a small nucleic acid sequence-a primer-attached to the RNA strand for initiation. Usually, it is a small nuclear DNA sequence at the site of insertion that plays this role for reverse transcription of Alu, however, in the cytoplasm, a sequence like this is not present.
By considering Alu’s RNA structure and its sequence, the team proposed that retrotransposons could perform priming on their own-that is, the molecule tucks on its own. Only another RNA molecule found in the rodent gene had been observed to self-prime, however, not inside the cytoplasm. The findings of the study indicated that, definitely, Alu is able to self-prime, providing the strand from which the enzyme, reverse transcriptase, can commence the synthesis process. Ambati mentioned that this really offered the mechanistic elucidation behind this occurrence inside the cytoplasm where nothing is present to prime it.
The Bridge Between Disease And Cytoplasmic DNA
Ambati and his team found Alu’s cytoplasmic synthesis while researching atrophic AMD (Age-related Macular Degeneration). 10 years back, they discovered that Alu RNA build-up activates cell death in a coating of the eye called RPE or Retinal Pigment Epithelium, and this condition is connected to untreatable diseases. From then onwards, a collection of findings has provided an explicit cellular image of the disease-for example, that Alu finally induces inflammation, the main symptom of several age-related problems, In this new project, the scientists demonstrated that Alu CDNA synthesized within the cytoplasm-not the one added into the genome-is vital for the toxicity behind atrophic AMD.
Due to the fact that Alu is a retrotransposon that depends on a reverse transcriptase in order to amplify its copy number, Ambati and his team proposed that NRTIs (Nucleoside Reverse Transcriptase Inhibitors)-meds now employed to cure retroviral disease-could obstruct Alu build-up. In 2014, the research team examined this proposal in rodent and human cells and discovered that these meds indeed stopped RPE degeneration, however, did so via their previously untold anti-inflammatory characteristics. In their recent study, the team proved that NRIs additionally block cytoplasmic cDNA synthesis of RPE cells, highlighting NRTI’s ability to hinder AMD on both the fronts-by intercepting transcription and by opposing inflammation.
As a result of the solid proof of NRTI’s effectiveness in stopping RPE degeneration in vitro, and since these meds have been utilized clinically for many years, the scientists thought of researching deeply within four distinct health insurance databases present in the US. They intended to examine if the application of NRTIs for other causes could have had the accidental profit of lowering the prevalence of atrophic AMD. Their assessments, comprising about 35 million adults, showed a lowered susceptibility of around 40% for atrophic AMD incidence in individuals consuming NRTIs.
Ambati, Inflammasome Therapeutics’ cofounder, a firm aimed at devising cures for degenerative diseases, asserted that finally, a prospective and randomized study is required to check whether the meds lower the development of atrophic AMD. as NRTIs are harmful, his team has devised altered forms, referred to as Kanuvudines, which are similarly effective, yet safer, in animal models and cell culture. According to Ambati, the firm will probably begin performing clinical trials with Kanuvudines this year itself.
Vera Gorbunova from the University of Rochester, who wasn’t involved in the study proclaimed that this is a very propitious path. She added that NRTIs could be considerably utilized as a therapy for numerous illnesses pertaining to inflammation. She researches aging and, as belonging to a group with whom Ambati has partnered, has found the cytoplasmic build-up of L1 cDNA in aged rodent cells. Her group has, in the past, reported the unknown mechanism behind the cytoplasmic cDNA formation, and mentioned that reverse transcription within the cytoplasm is one possible elucidation.
She said that when they issued that paper, they had to really struggle with the reverse transcription dogma that was hypothesized few years back-that it occurs inside the nucleus and that it employs genomic DNA, which it likely does when things go smoothly. However, when it is downhill, reverse transcription could possibly occur in the cytoplasm. She added that she is happy to see this proof.
Kang Zhang, an ophthalmologist from the Macau University of Science & Technology, who has partnered with Ambati and reviewed his recent paper before, reported that the discovery of cDNA being produced in the cytoplasm may also be significant to other diseases like cardiovascular disease, neurodegeneration, and cancer. There are numerous things occurring in the cytoplasm, This discovery has facilitated researchers like him to additionally focus on few phenomena that they couldn’t discern and also couldn’t elucidate. He added that it is quite exhilarating. This is a revolutionary change.
First Evidence of DNA Synthesis In the Cytoplasm




Hi guys, unfortunately the headline is incorrect – two groups showed L1-mediated DNA synthesis in the cytoplasm in mice and cells more than a year ago – Sedivy and Gorbunova in papers in Nature and Molecular Cell