Regulatory Affairs Career Roadmap: How to Enter and Grow in a Global RA Role
Hey, do you know that every medicine that you take today has reached you after a long approval process? Yes, and this entire process is managed by experts working in Regulatory Affairs careers.
The Regulatory Affairs professional makes sure every healthcare product is safe, effective, and legally approved before they reach patients. These products go through detailed reviews by regulatory authorities. The two most important global authorities are the U.S. Food and Drug Administration and the European Medicines Agency.
This is just the trailer of a bigger picture. Are you curious about this? Then let’s dive deep into this world. Together, we will take a look at the global regulatory affairs and everything related to them. Let’s begin.
What Is Regulatory Affairs?
Let’s fuel your curious mind with some basics first. Regulatory affairs (RA) is a field that is responsible for making sure that all the healthcare products meet all the regulatory requirements before entering the market. If you want to be one of them, your work includes managing approvals, maintaining complicated regulatory requirements, and communicating with regulatory agencies.
You can work in different sectors like pharmaceuticals, biotech, medical, cosmetics, and food safety.
Core Responsibilities of Regulatory Professionals
As a regulatory affairs professional, you will have to do the following tasks:
- You will be preparing regulatory submissions and documentation
- You have to communicate with regulatory authorities
- Along with it, you will be monitoring regulatory updates and policy changes
- Also, you have to make sure that compliance with international guidelines is met
- Most importantly, support product approvals and lifecycle management
A regulatory specialist works closely with research teams, clinical development groups, manufacturers, and quality departments. They bridge the gap between science, law, and business strategy. Without them, the healthcare products cannot legally enter the global markets.
Why is the Regulatory Affairs Career Growing?
As we know the healthcare industry is growing faster than ever. Every day, we have new therapies, biologics, digital tools, and personalized medicines. All of them need strict regulatory oversight.
Here are several factors that drive the demand for regulatory professionals:
-
Fast-moving Life Sciences Industry
With the rapidly changing world, pharma and biotech companies are launching products across the world. This requires experts who understand the international regulatory frameworks.
2. Growing complexity in regulations
The regulatory authorities regularly update their guidelines. They aim to improve patient safety and product quality. As an RA professional, you can help these companies in interpreting and implementing these changes.
3. Role of regulatory affairs in product approval and market entry
The regulatory strategy will determine how quickly the product will enter the market. The industry giants need experienced regulatory teams to accelerate approvals.
Due to these factors, regulatory affairs jobs have a high demand across the globe.
What education is needed for a regulatory affairs career?
To start your RA career, you need a strong educational foundation. If you are a graduate in Biotechnology, Life Sciences, or Biomedical Engineering, you are halfway there. These fields provide you with the scientific foundation required to understand the product and regulatory guidelines.
What specialized courses can help in regulatory affairs?
After completing your undergraduate degree, you can take up additional training courses. This will help you stand out from the crowd. One widely recognized credential is the Regulatory Affairs Certification (RAC) offered by the Regulatory Affairs Professionals Society. This certification will help you in globe job market.
You can also take up Biotecnika’s Global Regulatory Affairs course, which will help you gain hands-on experience with more benefits.
Key skills needed for success in regulatory affairs
To build a promising career in this field, you need a unique blend of technical, analytical, and communication skills.
| Skill | Why It Matters in a Regulatory Affairs Career |
| Scientific and Technical Knowledge | Understanding drug development, clinical trials, and manufacturing processes helps professionals prepare accurate regulatory submissions. |
| Regulatory Intelligence | Keeping track of updates from agencies such as the U.S. Food and Drug Administration, European Medicines Agency, and Pharmaceuticals and Medical Devices Agency helps ensure compliance with global regulations. |
| Communication and Collaboration | Regulatory professionals work with scientists, clinicians, legal teams, and regulatory authorities, so clear communication is essential. |
| Project Management | Managing regulatory submissions requires coordinating timelines, documentation, and teams across multiple departments. |
Regulatory Affairs Career Roadmap
The regulatory profession offers a structured career path. Most professionals move through several stages as they gain experience.
Typical Career Progression in Regulatory Affairs
| Career Level | Job Titles | Key Responsibilities | Experience Required |
| Entry Level | Regulatory Assistant, Regulatory Coordinator | Document preparation, regulatory data management, and support submissions | 0–2 years |
| Mid Level | Regulatory Affairs Associate, Regulatory Specialist | Prepare submissions, communicate with regulatory agencies, and support approvals | 2–5 years |
| Senior Level | Senior Regulatory Specialist, Regional Regulatory Lead | Develop regulatory strategies, manage submissions across regions | 5–10 years |
| Leadership | Regulatory Affairs Manager, Director, VP Regulatory Affairs | Lead global regulatory strategy, manage teams, and guide product approvals | 10+ years |
This progression highlights how a regulatory affairs career evolves from operational roles to strategic leadership.
Industries that hire regulatory affairs professionals
Regulatory affairs specialists are needed across several life sciences sectors.
| Industry | Role of Regulatory Affairs |
| Pharmaceutical Companies | Manage drug approvals and regulatory submissions for new medicines. |
| Biotechnology Firms | Support regulatory strategy for biologics and advanced therapies. |
| Medical Device Companies | Ensure devices meet safety and regulatory standards before market entry. |
| Contract Research Organizations (CROs) | Provide regulatory support and submission services for multiple clients. |
| Regulatory Consulting Firms | Offer regulatory strategy and compliance guidance for global markets. |
How to Get Your First Regulatory Affairs Job?
You can start your regulatory career; you need both educational and practical experience. To ease your journey, here is a roadmap.
-
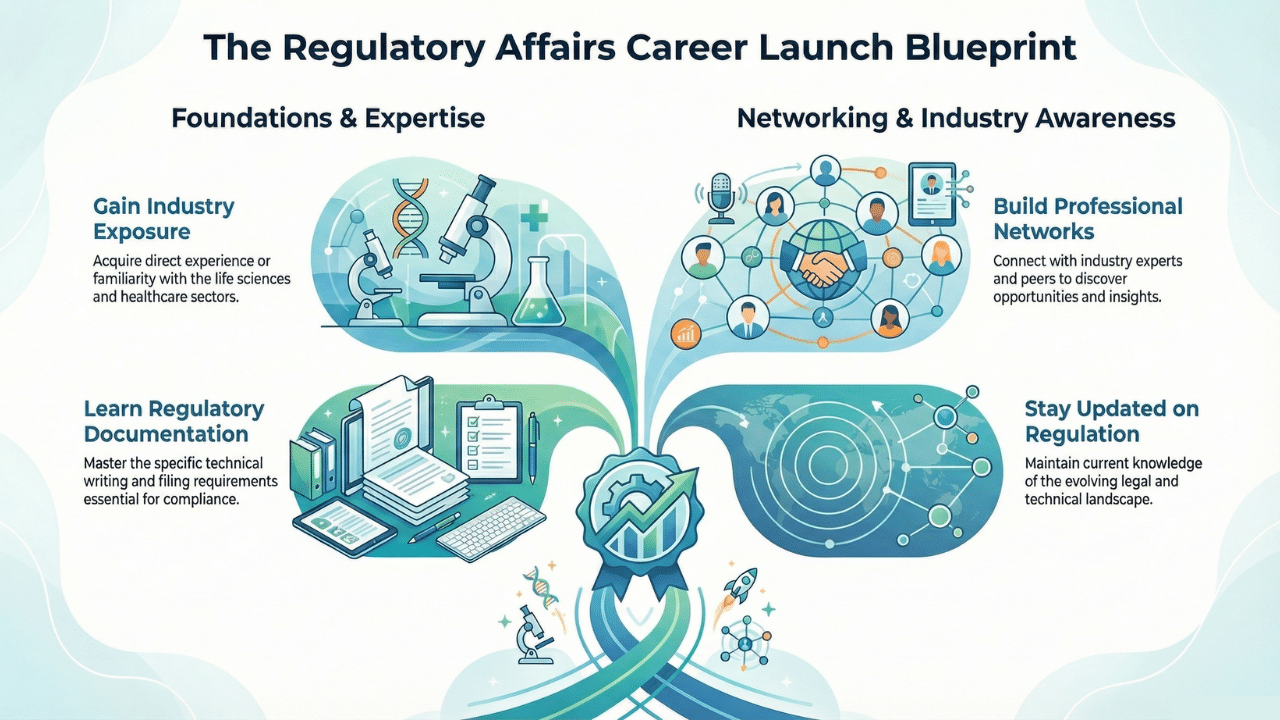
Gain Industry Exposure
After your degree, you can take up internships or entry-level roles in pharmaceutical companies. Also, the CROs provide valuable hands-on experience.
-
Learn Regulatory Documentation
The most important part you regulatory career is documentation. You have to understand regulatory formats. The important ones are the Common Technical Document (CTD), and electronic submissions are highly valuable.
-
Build Professional Networks
You can attend the conferences and seminars to gain industrial insights. This will also help in building your professional network with regulatory experts and hiring managers.
-
Stay Updated on Regulations
You must follow updates from global agencies. It will help you in understand changing regulatory requirements.
Benefits of choosing a regulatory affairs career
A regulatory affairs career offers several advantages:
- This field has a strong global job demand
- You will find many opportunities to work with international regulatory agencies
- The field offers competitive salaries and career growth
- You can directly contribute to public health and patient safety
Regulatory professionals, you will play a vital role in bringing safe and effective healthcare products to patients around the world.
Conclusion
The life science industry cannot function without this regulatory expertise. Every new product needs a strict regulatory evaluation before reaching the public. For someone like you who has a strong scientific background, this career provides a meaningful and promising opportunity.
With the right courses and practical experience, you can start your global career. You need to stay updated with the latest international regulations to stay ahead of the rest. Across the different industries, the demand for experts in global regulatory affairs and specialized regulatory affairs jobs will only increase.
If you are passionate about science, policy, and global health, then regulatory affairs offers you unique opportunities that can change your career path.



