The mechanism for the Division of Artificial Cells
A novel and generic mechanism for the division of artificial cells into two daughter cells uncovered by Max Planck scientists.
The amazing ability of live cells to divide themselves into two daughter cells is the reason for the success of life on the Earth. The outer cell membrane during this division process must undergo a series of morphological transformations that ultimately lead to fission of the cell membrane. By anchoring low densities of proteins to the artificial cell membranes, unprecedented control over these shape transformations and the resulting division process has been achieved by the scientists at the Max Planck Institute of Colloids and Interfaces, Potsdam, and at the Max Planck Institute for Polymer Research, Mainz.
The artificial cells are bounded by a single lipid membrane, which between the interior and exterior aqueous solution, provides a robust and stable barrier. Having the size of a typical animal cell, giant lipid vesicles are provided to these artificial cells. A crucial feature of cell membranes is this compartmentalization. Artificial cells with a wide membrane neck remain stable for days and weeks while when the neck has closed down the membrane, the artificial cell divides into two daughter cells as this closing of the membrane generates a constriction force onto this neck that cleaves the neck and divides the cells.
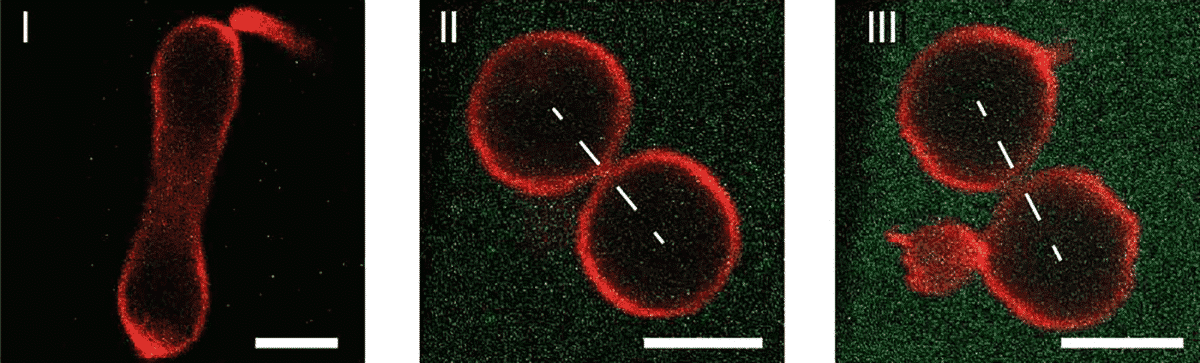
The researchers around Reinhard Lipowsky identified the novel mechanism, by which this constriction force can be controlled in a systematic manner, in addition to demonstrating the division of artificial cells. In order to do this, they designed the membranes by exposing the outer leaflets to a variable concentration of protein so that the inner and outer leaflets differ in their molecular composition. A preferred or spontaneous curvature that determines the shape of the artificial cells is generated by this asymmetry between the two leaflets. Moreover, the spontaneous curvature generates a local constriction force that leads to the division of these cells once a closed membrane neck has been formed.
Thus, surprisingly, the artificial cells’ complete division is driven by the membranes’ mechanical properties: From the asymmetry of the bilayer membranes, the force that divides the membrane neck arises directly.
For the division of artificial cells, a simple and generic mechanism has been identified in this way. By using different types of proteins, it has been demonstrated explicitly that this mechanism does not depend on the precise nature of the molecular interactions that generate the bilayer asymmetry and the associated curvature. On the artificial cell membranes, there was ample space left for other proteins to be accommodated as the used density of the membrane-bound proteins was rather low. Thus, a versatile and promising module for the bottom-up approach to synthetic biology is provided by the membrane-protein systems introduced here. Additionally, there is a new light shed on cell division in vivo through the division process of artificial cells described here.
The first author of the study, Jan Steinkühler explains, “Without the known protein machinery, certain bacteria can also divide. There is already a theory that in the latter division processes, membrane mechanics might be playing an important role. The possibility of mechanically controlled cell division is demonstrated in our study.”
Author: Prathibha