Molnupiravir- World’s First Antiviral Drug For COVID-19
To address the WHO’s (World Health Organization) increasing demand for vaccine equality in low-income and poor countries, a novel pill has been formulated. The pharma company, Merck, has requested immediate use approval for its oral drug, a pill that lowers death and hospitalization up to 50%.
Clinical studies of the drug, molnupiravir indicated that it lowered fatality counts and hospitalization risks when administered to high-risk individuals during the start of their infections. If FDA (Food & Drug Administration) authorizes the drug in the United States, Merck’s pill will be the first to get authorization amongst numerous other vaccines against the life-threatening SARS-CoV2.
Merck announced that it aims to increase its therapeutic production to 10 million courses with additional doses in the coming years. A few months back, Merck & the US government signed a procurement contract, under which it will provide around 1.7 million molnupiravir courses after the authorization of the pill.
The company stated that Merck has entered into purchase & supply contracts for the pill with other governments around the world. They also added that regulatory approvals are still pending and are now in discussions with international federal bodies.
What is molnupiravir?
Molnupiravir is a well-studied oral pill consisting of ribonucleoside that halts the replication of coronavirus, thereby preventing infection. Molnupiravir was developed by the DRIVE (Drug Innovations at Emory), a non-profit biotech firm which is associated with Atlanta’s Emory University. Currently, the drug is being manufactured by Merck together with Ridgeback Biotherapeutics.
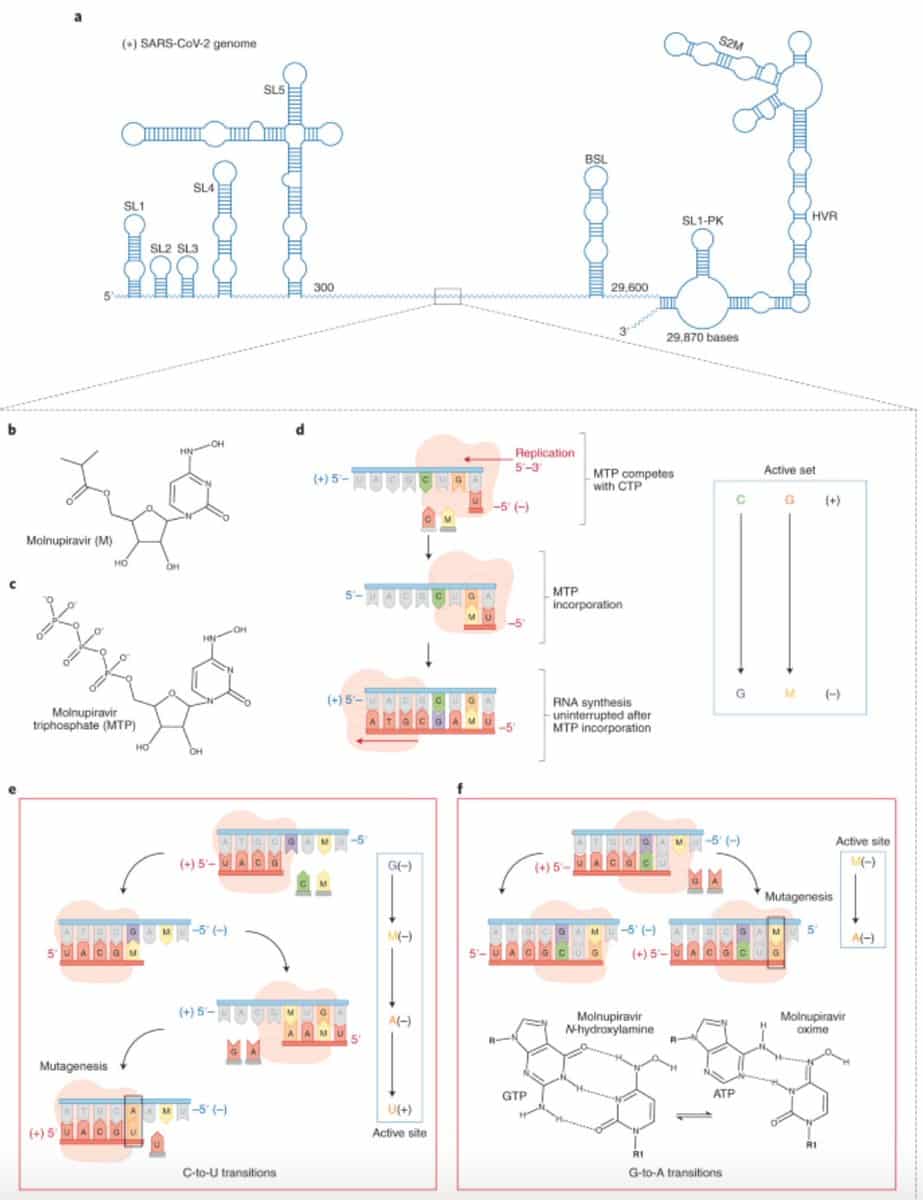
The drug triggers coronavirus to replicate its genetic content. Nonetheless, the drug induces errors in the genetic code when the virus begins the replication process. Additionally, this pill is being studied for a placebo-controlled, double-blind, randomized, multicenter, global Phase III trial, which is examining its safety & efficacy in preventing COVID-19 transmission in households.
Ridge Biotherapeutics’ CEO, Wendy Holman mentioned that due to the alarming rate of viral transmissions and the therapeutic limitations like restricted healthcare access & infusion therapy, therapeutic measurements against COVID-19 that can be conveniently used at home rather than hospitals are of imperative need.
What is the duration of the treatment?
Individuals who take Molnupiravir-world’s first antiviral pill have to complete a course that includes the daily consumption of two pills for a period of five days. Totally, individuals have to consume 40 capsules.
What is the efficacy of Molnupiravir-world’s first antiviral pill?
The recent findings from Phase 3 studies indicated that the drug was capable of lowering the fatality risk as well as the risk of hospitalization in half. Among the groups studied, 14% of placebo group volunteers were hospitalized or dead and 7% of individuals belonging to the group who received the medication were hospitalized with no fatalities.
Merck confirmed that the drug lowered the fatality & hospitalization risks across all major subgroups. Efficiency was not influenced by the underlying risk factors or symptom onset’s time. The firm declared that according to the volunteers with accessible viral sequencing data (nearly 40% of volunteers), molnupiravir showed uniform efficiency for all viral variants- Mu, Delta, and Gamma.
The clinical study was performed at over 170 planned locations across the world including the US, the UK, Ukraine, Taiwan, Sweden, Spain, South Africa, Russia, Poland, Philippines, Mexico, Japan, Italy, Israel, Guatemala, Germany, France, Egypt, Colombia, Chile, Canada, Brazil, and Argentina.
What are the possible side effects of molnupiravir?
No detrimental side effects have been found from the drug among the volunteers. Nonetheless, there could be possibilities of mild headaches like the symptoms after vaccination against COVID-19.
Why Indian Pharma Companies Ended The Studies?
Even though Merck came up with positive clinical trial outcomes, two other Indian Pharma companies requested for ending the final trials of their generic forms of Merck’s antiviral orally administered experimental drug by the start of October 2021. Reuters stated that MSN Laboratories & Aurobindo Pharma Ltd didn’t rule out hospitalized individuals for their clinical trials, based on the trial documents, even though it wasn’t clearly indicated whether the studies included hospitalized individuals.
Merck’s studies followed the US FDA standards. According to this definition, the blood oxygen level for moderate COVID-19 is no less than 93%. Nonetheless, the same for India is 90 to 93%. Based on the website of the internal expert committee belong to the Indian drug regulator and the study documents, Reuters reported that MSN and Aurobindo are resuming their research in non-hospitalized individuals with mild COVID-19 infection.
The organization citing the sources announced that the drug has not demonstrated significant efficiency for moderate COVID-19 infections in trials conducted in India, though it showed promising results in mild infections.
Is this pill the only oral option?
Merck’s pill is not the only option. Pfizer is also researching oral medications for COVID-19 infections. Even though clinical study findings are yet to be declared, this alternative pill will most probably be available by 2022.
Molnupiravir-World’s First Antiviral Pill